Current Research
Cyanobacterial Biology and Biotechnology
Cyanobacteria (Fig. 1) are some of the simplest microbes capable of harnessing energy from sunlight and are among the most evolutionarily ancient organisms. Indeed, cyanobacterial biochemistry, acting over millions of years, radically altered the atmosphere and enabled life as we know it on Earth today. It seems therefore fitting that cyanobacteria could be an important component to help us address some of today’s urgent energy and atmospheric problems.
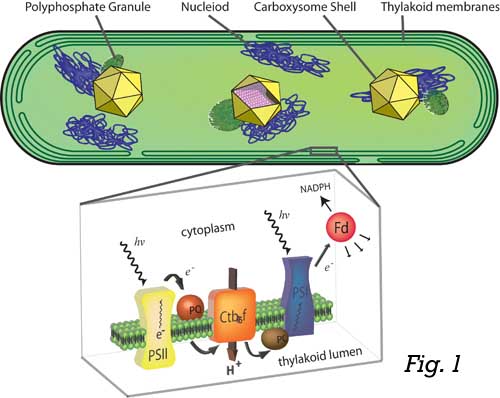 Our lab is interested in using engineering and synthetic approaches to examine the
biology and biotechnological application of these intriguing bacteria. Cyanobacteria
are thought to be the evolutionary root of all photosynthetic capacity found in algae
and plants, so they possess similar light gathering/processing machinery to more complex
organisms, including crop species. Yet, cyanobacteria possess the capacity to convert
solar energy into biomass with greater efficiency than land-based plants and can be
found thriving in much more extreme environments. Indeed, cyanobacteria are the base
of many ecosystems where plants cannot grow, including artic lakes, hot springs and
salty environments. Cyanobacteria therefore hold considerable promise as an efficient
platform for the sustainable production of economically important compounds in a manner
that could use marginal land and water supplies unsuitable for growing crops or supporting
diverse ecosystems.
Our lab is interested in using engineering and synthetic approaches to examine the
biology and biotechnological application of these intriguing bacteria. Cyanobacteria
are thought to be the evolutionary root of all photosynthetic capacity found in algae
and plants, so they possess similar light gathering/processing machinery to more complex
organisms, including crop species. Yet, cyanobacteria possess the capacity to convert
solar energy into biomass with greater efficiency than land-based plants and can be
found thriving in much more extreme environments. Indeed, cyanobacteria are the base
of many ecosystems where plants cannot grow, including artic lakes, hot springs and
salty environments. Cyanobacteria therefore hold considerable promise as an efficient
platform for the sustainable production of economically important compounds in a manner
that could use marginal land and water supplies unsuitable for growing crops or supporting
diverse ecosystems.
We are a young lab with prior experience engineering cyanobacterial strains that can convert solar energy into the biofuel hydrogen gas, or excrete bioindustrially-compatible sugars for downstream applications. Our aim is to continue to utilize traditional engineering and emerging synthetic biological tools to refine our capacity to compartmentalize and control the complex metabolism of cyanobacteria. These tools have allowed us to probe the flexibility of photosynthetic metabolism and generate strains with emergent properties that have broader implications for understanding dynamic feedback and regulation between metabolism and light gathering in green microbes and plants.
For example, we have recently designed a strain of cyanobacteria that can export a
significant fraction of the carbon it fixes (up to 85%) as sucrose. This approach
is promising as a potential alternative to land-based crops for sugar production (Fig. 2), as this level of productivity could exceed sugar output from sugarcane or corn
if it could be scaled. 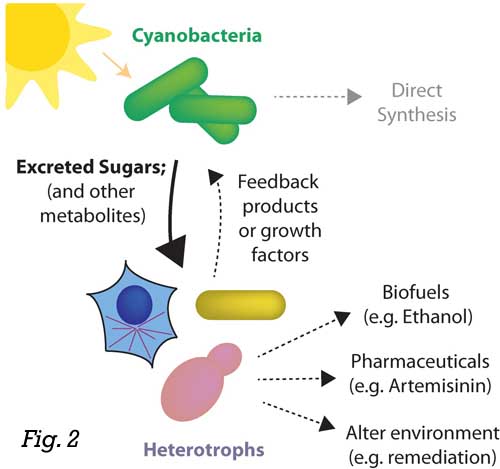
Furthermore, this system also allows us to place a novel metabolic burden on the cell
in a tunable fashion and investigate the response – an approach that can reveal
aspects of the basic physiology of the system. Counter-intuitively, we have found
that these cyanobacterial strains respond by upregulating their photosynthetic capacity
and increasing their CO2 fixation rate to help keep pace with the demand. Understanding
this phenomena, and other adaptive responses, could lead to a better understanding
of how to poise metabolism for maximal photosynthetic efficiencies and the most effective
biological sustainable solutions. Ongoing projects in the lab aim to contribute broadly
in the areas of alternative energy development, underst